Safer Meds
Secure medicine take-back programs. Pharmaceutical stewardship policies. Reducing medicine abuse & poisonings. Reducing pharmaceutical pollution.Learn About Drug Take-Back Laws in Washington State
Success! From a vision to pilot programs to bills to local laws to a state law to the first-in-nation comprehensive statewide drug take-back program financed and operated by the pharmaceutical industry!
The WA Secure Drug Take-Back Act was signed into law on March 22, 2018. An approved drug take-back program launched on November 21, 2020. Secure medicine drop boxes are now distributed across Washington in pharmacies, clinics, hospitals, and police stations. Prepaid mailers are available to residents upon request and will also be distributed by participating pharmacies.
Washington’s law is the most comprehensive pharmaceutical stewardship law in the nation. It requires the pharmaceutical industry to finance and coordinate a secure and convenient drug take-back program to make it easier for residents to safely dispose of leftover and expired medicines.
This legislative victory is the result of a multi-year policy and advocacy campaign involving a wide array of organizations, agencies, and individuals across the state.
The state law is modeled on and builds on the success of similar local Secure Medicine Return ordinances enacted in 7 counties in Washington State. The pharmaceutical industry’s MED-Project programs are operating in Clallam, King, Kitsap, Pierce, Skagit, Snohomish and Whatcom counties. The local laws will remain in force until 1 year after drug manufacturers begin operating an approved statewide drug take-back program under the state law. The local programs will then merge into a consistent statewide program serving all communities across Washington.
This page provides overview and summary information about the WA Secure Drug Take-Back Act and the local Secure Medicine Return ordinances. Contact me to learn how we achieved these policy successes!
About Margaret Shield. Principal, Community Environmental Health Strategies
Margaret has worked extensively on pharmaceutical stewardship legislation and drug take-back regulations. She was the lead policy staff for the King County WA Board of Health’s process to develop a Secure Medicine Return ordinance. As a consultant, she worked with local health agencies and health boards in Snohomish, Pierce, Whatcom, and Skagit counties to develop their policies. Margaret also played a leading role in the policy and advocacy campaign to pass the WA Secure Drug Take-Back Act. She is currently consulting on similar policies in other states.
WA Secure Drug Take-Back Act of 2018
Local Secure Medicine Return Ordinances in WA
The Secure Drug Take-Back Act – RCW 69.48; House Bill 1047 – sponsored by Representative Strom Peterson, passed the WA Legislature with strong bipartisan support and was signed into law by Governor Inslee on March 22, 2018. The law took effect on June 7, 2018 and a related rule took effect on August 1, 2019.
The law is a pharmaceutical stewardship policy that requires the pharmaceutical companies that manufacture medicines to finance and coordinate a convenient and secure collection system for consumers to properly dispose of unused medicines.
The WA State Department of Health has reviewed a drug take-back program proposal submitted by pharmaceutical manufacturers. The program will be operated by MED-Project. After requiring several revisions to address deficiencies, WA DOH accepted the MED-Project program proposal as meeting the law’s requirements on May 25, 2020. The official launch date of the MED-Project program was November 21, 2020. The program’s website is med-project.org/locations/washington/
Explore this section for summaries of the law, as well as more in-depth explanation of key parts of the law’s policy for convenience, financing, and implementation timing.
Resources
Drug Take-Back Options in WA (PDF, 2 pages) summarizes drug take-back programs in WA as of April 2018 and provides brief overview of new WA Secure Drug Take-Back Act
Brief Policy Overview of WA Secure Drug Take-Back Act (PDF, 1 page)
Implementation Timing in the WA Secure Drug Take-Back Act (PDF, 1 page)
Detailed Policy Overview of WA Secure Drug Take-Back Act (PDF, 34 pages) includes complete text of law, detailed policy summary, implementation timeline, and legislative section list.
03/22/2018 Media Release: Secure Drug Take-Back Act Becomes Law (PDF, 5 pages) media release from Rep. Strom Peterson and supporters of ESHB 1047; includes quotes from supporters and links to additional resources.
Resources used by supporters of the WA Secure Drug Take-Back Act during the 2018 legislative session:
WA Secure Drug Take-Back Act Handout (PDF, 2 page)
Supporters List (PDF, 1 page)
Resources provided on this site may not be used for commercial purposes, reused without attribution, or modified for reuse. Please link to this site for the latest updates to these resources.
Policy Summary of the WA Secure Drug Take-Back Act
The WA Secure Drug Take-Back Act has been codified as RCW 69.48.
Section numbers listed in this summary refer to the numbering in the final bill ESHB 1047.
A statewide drug take-back program for prescription and over-the-counter medicines must be financed and coordinated by pharmaceutical manufacturers selling medicines into WA. Sec. 3, 5, 9.
Medicine manufacturers design their program to meet the bill’s requirements and standards, and manage their costs for drop boxes, collection supplies, prepaid mailers, collection events, transportation, disposal, and promotion. In-kind contributions from pharmacies, clinics, hospitals, and law enforcement agencies that volunteer to host and staff secure drop boxes.
Estimated program costs to drug manufacturers are about 0.1% of annual medicine sales of $5.7 billion in WA.
Convenient access to secure medicine drop boxes at pharmacies, hospitals and police stations in all cities and towns. Sec. 6.
The manufacturers’ program must include any qualified pharmacy, hospital/clinic, or police station that volunteers to host a secure drop box as a collection site.
All collectors participate voluntarily.
At least 1 collection site provided in every city/town’s population area (defined as including a 10-mile radius around each city or town), plus 1 additional collection site for every 50,000 residents.
For islands and unincorporated areas outside population areas, collection site must be provided at every authorized collector open to the public, unless the collector is unwilling or unqualified.
In any areas underserved by collection sites, as determined by WA Dept. of Health and local health agencies, mailer distribution locations or periodic collection events must be provided for residents.
Prepaid return mailers provided on request to any resident and to any retail pharmacy that offers to distribute mailers. Sec. 6.
Local input on program services from health agencies, law enforcement & stakeholders. Sec. 5, 6.
Acceptance of all medicines used in the home, both prescription and over-the-counter, including legally prescribed controlled substances like OxyContin, Vicodin, and stimulants. Sec. 2.
With logical exemptions for personal care products regulated as drugs, like lip balm, toothpaste, and sunscreen.
Public education and outreach on safe medicine storage and using the drug take-back program. Sec. 7.
Manufacturers must provide a website, toll-free number, and distribute educational materials.
State agencies and pharmacies help promote the program. Local governments are encouraged to promote.
Secure drug collection and handling procedures per the DEA’s Rule, and all other applicable federal & state laws and regulations. Sec. 5, 6.
Environmentally sound disposal of collected medicines at properly permitted high temperature incineration facilities, using hazardous waste facilities when feasible or using a large municipal solid waste combustor (e.g. WTE) as recommended by the EPA. Sec. 8.
WA Department of Health oversight for security, safety, and compliance. Sec. 5, 7, 10, 11, 12, 19, 20.
WA DOH will review and approve the manufacturers’ drug take-back program plan. DOH oversight includes monitoring the program, reviewing annual program reports, and conducting periodic public awareness surveys.
Program evaluation will be conducted by WA Department of Health, an academic institution, and by the WA Poison Center to assess the program’s impact on resident awareness and behavior, rates of medicine abuse and poisonings, and proper disposal of drugs to prevent pharmaceutical pollution.
Much of WA DOH’s oversight costs will be recovered by a fee on approved drug take-back program(s).
Local Secure Medicine Return ordinances remain in effect until 12 months after the statewide drug take-back program begins operations. Sec. 16.
The state bill’s requirements are very similar to local ordinances enacted in a number of WA counties. Manufacturers must provide local programs under these ordinances until 1 year after the statewide program is launched.
Otherwise local laws regulating drug take-back programs are preempted under the statewide law.
The Act sunsets in January 2029. The Legislature can review and reauthorize the drug take-back program.
"Convenience Standard" for the WA Secure Drug Take-Back Act
Effective pharmaceutical stewardship policies have specific performance standards and requirements for the drug manufacturers’ take-back program. The “convenience standard” defines the number and types of collection services that must be available to residents. WA’s law defines a multi-part convenience standard with both general and specific requirements to ensure access for residents throughout the state. See Section 6 of ESHB 1047 for complete language. (The WA Secure Drug Take-Back Act has been codified as RCW 69.48.)
General convenience standard: The collection system must be safe, secure, operate on a year-round basis and provide equitable and reasonably convenient access for residents across the state. All “covered drugs”, including legally prescribed controlled substances, must be accepted by all collection methods. Must give preference to locating secure drug drop boxes at pharmacies, clinics, hospitals, and law enforcement agencies. Sec. 6(3)(a and b)
“Opt-in” for qualified collectors: Any retail pharmacy, hospital or clinic with an on-site pharmacy, or law enforcement agency that volunteers to host a secure drug drop box must be included in the manufacturer’s drug take-back program, within 90 days of volunteering. Sec. 6(1)(c)
Manufacturers may also include other DEA authorized collectors in their program, like narcotic treatment programs or specific types of long-term care facilities. Sec. 6(1)(d)
There is no limit to the number of drug drop boxes that manufacturers must finance under this “opt-in” requirement. All DEA authorized collectors participate voluntarily. Sec. 6(1)(b)
Minimum number of collection sites in cities & towns: The manufacturers’ drug take-back program must provide a minimum of 1 collection site in each population center (defined as a city or town and the unincorporated area within a 10-mile radius) plus 1 additional collection site for every 50,000 residents of the city or town in the population center. Sec. 6(3)(c)(i)
Collection sites on islands an in unincorporated areas: On islands and in areas outside population centers (defined as a city or town and the unincorporated area within a 10-mile radius), the drug take-back program must provide a drop box at the site of every potential authorized collector that is regularly open to the public, unless that site is unwilling or unqualified. Sec. 6(3)(c)(ii)
Services for any areas underserved by drop boxes: In any areas of the state that are underserved by collection sites, as determined by WA DOH in consultation with the local health jurisdictions and community, the manufacturers’ drug take-back program must provide mailer distribution locations or collection events. Local stakeholders have input on the number/locations of mailer distribution locations and frequency of collection events. Sec. 6(3)(f)
Mail-back services: Any resident can request a prepaid, preaddressed return mailer via the program’s website or toll-free phone number. Any retail pharmacy that offers to distribute mailers must be provided with mailers to give to customers upon request. Sec. 6(e)
Financing Mechanism of the WA Secure Drug Take-Back Act
Section numbers listed in this summary refer to the numbering in the final bill ESHB 1047.
Manufacturers pay for and operate the drug take-back program, controlling their own funds.
- The law’s pharmaceutical stewardship policy requires drug manufacturers selling medicines in or into Washington State to participate in an approved drug take-back program. Medicine manufacturers design their own program(s) and manage their own finances to meet the bill’s standards for a system of secure medicine drop boxes, prepaid return mailers, and public education about safe medicine storage and secure disposal. Sec. 3, 5, 9.
- Drug manufacturers are responsible for all administrative and operational costs of their drug take-back program, including but not limited to:
- secure collection receptacles;
- collection and transportation supplies;
- prepaid return mailers;
- law enforcement staff time for collection events, if utilized as a collection method;
- transportation and final disposal of collected medicines; and
- costs of required promotion and outreach. Sec. 9.
- A point-of-sale fee or point-of-return fee is not allowed. Manufacturers can pass costs through the supply chain to purchasers of drugs, which include healthcare providers, insurance companies, and consumers. Sec. 9.
- Manufacturers determine among themselves how to apportion the program costs. Sec.5.
- “Covered manufacturer” is defined in the bill as pharmaceutical companies that produce, prepare, and process drugs, similar to the definitions of “manufacture” of drugs used in WA statute (RCW 18.64.011) and by the FDA. “Covered manufacturer” does not include retail pharmacies with a store label drug, private label drug distributors, or drug repackagers provided that the manufacturer of the drug is identified by those entities. Sec. 2.
Pharmacies, hospitals, and other collectors provide in-kind support by hosting secure drop boxes and assisting with public education. Pharmacies, clinics, hospitals, law enforcement agencies, and other DEA authorized collectors volunteer to host and staff secure drop boxes, and assist with consumer education. Sec. 6, 7.
Agency oversight costs are recovered. Costs of WA Department of Health oversight are largely recovered through an annual fee assessed on each approved drug take-back program. Fees must be used for actual costs of administration, oversight, and enforcement. Starting in 2020, annual department fees are capped at 10% of the manufacturers’ drug take-back program costs. Sec. 12.
The estimated cost of drug take-back program to pharmaceutical manufacturers is only about 0.1% of annual medicines sales in Washington State. That’s investing about 1 penny for public safety from every $10 in medicine sales.
Implementation Timing for the WA Secure Drug Take-Back Act
Most deadlines in the law are relative to the effective date, or to the fixed deadline of July 1, 2019 for submission of program proposal(s) from manufacturers. Please view these dates as estimated until confirmed by the WA State Department of Health.
The WA Secure Drug Take-Back Act has been codified as RCW 69.48. Section numbers listed in this summary refer to the numbering in the final bill ESHB 1047.
June 7, 2018 Effective Date; 90 days after Sine Die of 2018 Legislature on March 8, 2018.
Local jurisdictions preempted from mandating drug take-back by pharmacies, clinics, hospitals, and law enforcement. Sec. 16.
Enacted county laws requiring drug manufacturers to provide drug take-back are grandfathered and may be enforced until 12 months after an approved statewide drug take-back program begins operations. Sec. 16.
June 2018 ? – ? WA State Department of Health (DOH) Rule-making. Time frame TBD. Sec. 18.
By Sept. 5, 2018 (90 days after effective date): Drug wholesalers must provide lists of drug manufacturers to DOH. Retail pharmacies, private label distributors, and repackagers that sell a drug under their own label must identify manufacturers to DOH. Sec. 4.
By March 2, 2019 (120 days before Program Proposal due date of July 1st): The Program Operator for a covered manufacturers’ program must notify potential authorized collectors of opportunity to host a secure drug drop box. Sec. 6.
By July 1, 2019 Drug Take-Back Program proposal(s) due from covered manufacturers to DOH. Sec. 5
By July 1st: DOH determines its administration, oversight, and enforcement costs and sets annual fee from each program operator. DOH collects fee from program operator(s) by Oct. 1, 2019 and annually thereafter. Sec. 12.
By Oct. 29, 2019 (within 120 days after submission): DOH must approve or reject Program proposal(s). Sec. 5.
If proposal rejected, program operator has 90 days to resubmit.
If proposal accepted, the program operator must initiate operation of the Drug Take-Back program within 180 days. Sec. 5.
May 2020 Potential earliest starting month for an approved statewide Drug Take-back Program if the first Program proposal from covered manufacturers is accepted by DOH. Sec. 5. [Update: The actual launch of an approved program occurred on 11/21/2020 after approval of revised Program proposals.]
12 months after start of approved statewide Drug Take-Back Program: Grandfathered county laws are preempted. Program Operator(s) must work to integrate each local program with their approved statewide program during the 1 year period. Sec. 16
Annual Reporting by Covered Manufacturers. Sec. 10.
- 30 days after each annual period of operation, Program Operator must submit collection report on amount of medicines from each collection site.
- Annual reports from Program Operators due July 1st after first full year of program operation, and on July 1st annually thereafter. Report includes pounds of medicines collected by each collection method; description of education activities, program expenditures by category, and evaluation of program’s success in meeting goals for collection amounts & public awareness.
Reporting and Periodic Public Awareness Surveys by WA DOH.
- 30 days after program proposal approved, DOH submits limited first report to the Legislature. DOH reports to Legislature are due next Nov. 15th and biennially thereafter. Sec. 19.
- DOH must conduct a public awareness survey after first full year of program operations and every 2 years thereafter; and may require the program operator to modify promotion and outreach to improve public awareness. Sec. 7.
State Rulemaking Process
The Washington State Department of Health (WA DOH) has completed its rule-making process to implement the WA Secure Drug Take-Back Act passed by the 2018 Legislature. The law clearly defines key goals and standards that the drug take-back program must achieve, including services that must be provided across the state. The rule clarifies some details and processes. The final rule is Chapter 246-480 WAC.
WA DOH’s rule-making process was initiated in June 2018 and completed in just over one year. Public workshops were held during August and September and public comments were accepted at these workshop and on a draft rule. WA DOH issued a final draft rule, a CR-102, in early 2019 and adopted the final rule on July 1, 2019. The rule took effect August 1, 2019.
Additional information is available on WA DOH’s Drug Take-back Program website: https://www.doh.wa.gov/SafeMedReturn
Want to understand pharmaceutical stewardship policies? Or get started on legislation?
- explaining the pharmaceutical stewardship policy approach to comprehensive and sustainably financed programs.
- providing comprehensive background and analysis on key decision points and common questions.
- examining policy options and customizing legislative language for your state or local jurisdiction
- addressing medicine take-back operations and key regulatory issues in the context of policy development.
- developing an effective policy communications and outreach strategy.
Learn more about my Services
In response, roughly 400 pharmaceutical manufacturers are participating in and financing the MED-Project LLC stewardship organization that is operating approved drug take-back programs under the local laws.
As of December 2018, drug manufacturers’ are operating their MED-Project program in King, Kitsap, Pierce, Snohomish, and Whatcom counties. Secure drug drop boxes are provided in participating pharmacies, clinics, hospitals, and law enforcement agencies. Prepaid return mailers are also available for residents with limited mobility.
In Clallam and Skagit counties, drug manufacturers must submit stewardship plans during 2018 according to deadlines, with anticipated program starts in the first half of 2019 depending on the length of the plan review and approval process.
The recently passed WA Secure Drug Take-Back Act was modeled on these successful local ordinances. Requirements of the state law and the local laws are very similar. Manufacturers must comply with the local laws until 1 year after they begin operating an approved statewide drug take-back program under the state law. The manufacturers’ program will then merge into a consistent statewide program providing secure drug take-back options in all communities across Washington.
History of Local Secure Medicine Return Ordinances in WA State
The Board of Health of King County passed Washington’s first local law in July 2013. King County’s law is similar to the nation’s first pharmaceutical stewardship law passed by the Board of Supervisors of Alameda County, CA in July 2012.
Alameda County and King County were each sued in federal court, unsuccessfully, by pharmaceutical industry associations. Both counties voluntarily extended their deadlines for stewardship plan submission as the Alameda case worked through the courts. The Northern California District Court and the Ninth U.S. Circuit Court of Appeals each upheld Alameda County’s ordinance, dismissing the industry’s claim of a violation of the dormant Commerce Clause. After the Ninth Circuit ruling in September 2014, both counties resumed implementation deadlines. A petition from the pharmaceutical associations to the U.S. Supreme Court to review the Ninth Circuit ruling was denied in May 2015.
After resolution of the unsuccessful industry lawsuit and with the DEA’s 2014 Rule for Disposal of Controlled Substances in effect, additional counties in Washington and California began adopting pharmaceutical stewardship ordinances. In mid-2016, Snohomish County, WA enacted the second similar law in Washington State. The Snohomish ordinance is very similar to King’s, with some refinements to further align language with the DEA’s Rule, an enhanced service convenience goal, shortened implementation timelines, and expanded promotion requirements. Kitsap and Pierce counties enacted their ordinances in December 2016, followed by Clallam and Whatcom in late 2017, and Skagit County in February 2018.
Resources
Secure Medicine Return Laws in WA State (PDF, 2 pages)
summarizes implementation status of county-level pharmaceutical stewardship laws; lists number of secure drop boxes as of January 2019.
Drug Take-Back Options in WA (PDF, 2 pages) summarizes drug take-back programs in WA as of April 2018 and provides brief overview of new WA Secure Drug Take-Back Act
Resources provided on this site may not be used for commercial purposes, reused without attribution, or modified for reuse. Please link to this site for the latest updates to these resources.
King County
- King County Board of Health Chapter 11.50 Passed June 20, 2013; Effective July 20, 2013.
- Approved Stewardship Plan
- King County’s program website: kingcountysecuremedicinereturn.org/
- Drug Manufacturers’ MED-Project Program Website: med-project.org/locations/king-county/
Implementation Status: Manufacturers’ MED-Project program operating since November 2016.
Collection Data: During 2017, the first program year, 39,197 pounds of medicines were securely collected and safely destroyed.
Source: MED-Project 2017 Annual Report to King County (PDF on Google docs)
As of January 2019: About 115 secure medicine drop boxes. More than 75% of drop boxes are at pharmacies, clinics, hospitals and the rest are at law enforcement agencies. Authorized collectors may join the program at any time, so the number of drop boxes is still increasing.
Prepaid mailers distributed at 17 libraries and some town halls.
Mailers are available to home-bound residents & their service providers upon request via website or phone.
Before implementation of the law: King County had only 3 pharmacy drug drop boxes and 10 police drop boxes in smaller towns.
Snohomish County
- Snohomish Health District Code Chapter 2.80 Passed June 14, 2016; Effective July 15, 2016.
- Approved Stewardship Plan
- Snohomish Health District program website: http://snohd.org/204/Pharmaceutical-Stewardship
- Manufacturers’ MED-Project Program Website: med-project.org/locations/snohomish/
Implementation status: Manufacturers’ MED-Project program operating since August 2017.
As of January 2019:
- 37 secure medicine drop boxes, roughly half at pharmacies, clinics, hospitals and half at law enforcement agencies. Authorized collectors may join the program at any time, so the number of drop boxes is still increasing.
- Prepaid mailers are available to home-bound residents & their service providers upon request via website or phone.
- Coming in near future – prepaid return mailers for pre-filled injector products.
Before implementation of the law: only 2 pharmacy drop boxes and 25 law enforcement drop boxes without stable funding.
Kitsap County
- Ordinance 2016-02: Secure Medicine Return Regulations Passed and effective Dec. 6, 2016.
- Kitsap Public Health District’s program website: kitsappublichealth.org/information/medicine_return.php
- Drug Manufacturers’ MED-Project Program Website: med-project.org/locations/kitsap-county/
Implementation Status: Manufacturers’ MED-Project program began rolling out in March 2018.
As of January 2019:
- 12 secure medicine drop boxes at 8 pharmacies or clinics and 4 law enforcement agencies.
- Prepaid mailers are available to home-bound residents & their service providers upon request via website or phone.
- Prepaid mailers are also available to residents for iodine-containing medications.
Before implementation of the law: 2 drop boxes at Sheriff’s offices and 1 pharmacy drop box.
Pierce County
- TPCHD Environmental Health Code Chapter 7 Passed Dec. 7, 2017; Effective Dec. 8, 2017.
- Approved Stewardship Plan and conditional approval letter
- Tacoma-Pierce County Health Department’s program website: www.tpchd.org/healthy-places/waste-management/secure-medicine-return
- Drug Manufacturers’ MED-Project Program Website: med-project.org/locations/pierce/
Implementation status: Manufacturers’ MED-Project program began rolling out in April 2018.
As of January 2019:
- 38 secure medicine drop boxes, roughly half at pharmacies or clinics and half at law enforcement agencies. More boxes being installed.
- Prepaid mailers are distributed from 2 locations and available to home-bound residents & their service providers upon request via website or phone.
- Prepaid mail-back packages are also available to all residents for inhalers, pre-filled injector products, and iodine-containing medications (each product type returned separately).
Before implementation of the law: drop boxes at 4 pharmacies and 17 law enforcement locations.
Clallam County
- Board of Health Ordinance 6 Passed and effective Nov. 21, 2017
Implementation status: stewardship plan deadline in May 2018.
Whatcom County
- Whatcom County Code Chapter 24.15 Passed and effective Dec. 5, 2017.
- Whatcom County Health Department’s program website: https://www.whatcomcounty.us/2652/Secure-Medicine-Return
- Drug Manufacturers’ MED-Project Program Website: med-project.org/locations/whatcom-county/
Implementation status: Manufacturers’ MED-Project program launched in November 2018. The MED-Project stewardship plan was conditionally approved August 31, 2018. In November 2018, the MED-Project program began installing secure drop boxes.
As of January 2019:
- 6 secure drug drop boxes at 5 drug stores and 1 police department. More drop boxes being installed. A local pharmacy drop box program expected to join manufacturers’ MED-Project program soon, and expand their collections to include controlled substances.
- Prepaid return mailers available upon request for homebound residents and providers of services to those residents, such as hospice providers.
- Prepaid mail-back packages are also available to all residents for inhalers and pre-filled injector products (each product type returned separately).
Skagit County
- Skagit County Code Title 12, Chapter 20 Passed and effective Feb. 21, 2018
- Skagit Public Health Department’s program website: https://www.skagitcounty.net/Departments/Health/medicinereturn.htm
Implementation status: stewardship plan deadline in August 2018. Anticipated program start in February 2019.
Exploring a local pharmaceutical stewardship policy?
- public health and environmental concerns with leftover medicines.
- options for secure collection, transport, and environmentally sound disposal.
- key regulatory considerations.
- costs and funding considerations.
- policy and legislative approaches for comprehensive and sustainable programs.
Learn more about my Services
It’s a Go! WA’s Statewide Pharmaceutical Stewardship Program Launched!
Washington State Celebrates the Official Start of the First Statewide, Comprehensive Drug Take-Back Program Operated by the Pharmaceutical Industry! A fitting subtitle is "good things come to those who organize and persist". It took many years of educating,...
Pharmaceutical Stewardship Passes in Oregon, 4th Law in Nation!
A fourth state Legislature has voted overwhelmingly for a pharmaceutical stewardship, a.k.a. extended producer responsibility, approach to providing convenient drug take-back services to all residents. After many years of discussion on the issue, the Oregon...
Another look at medicine disposal products: performance and effectiveness?
My latest dive into assessing whether medicine disposal products provide a safe, effective, and environmentally sound way to dispose of pharmaceuticals is now out in report form. Medicine Disposal Products: an overview of products & performance questions examines...
National Association of Counties article on WA drug take-back successes!
Thanks to NACo for helping share the news about successful pharmaceutical stewardship laws in Washington State. NACo County News published my article explaining how local policies served as innovative models and drove the adoption of WA's statewide law. ...
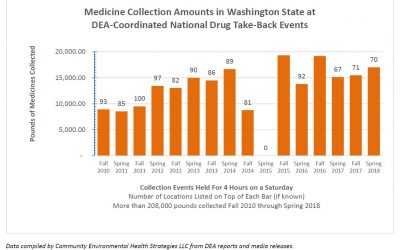
15th National DEA Drug Take-Back Event Captures Almost 1 Million Pounds!
In just 4 hours on Saturday April 28th, people across the U.S. returned almost 1 million pounds of leftover and expired medicines for safe disposal. The 15th National DEA Drug Take-Back Day netted 949,046 pounds of drugs or 475.5 tons! Complete results of the...
Secure Drug Take-Back Act Becomes Law in WA!
Proponents of pharmaceutical stewardship in Washington State are celebrating a new law to help reduce medicine misuse, poisonings, overdoses, and that will also help reduce pharmaceutical pollution - the WA Secure Drug Take-Back Act. This...
What About Medicine Disposal Products? Effective? Safe? Worth the Price?
New products in pouches and bottles are being marketed to consumers with claims that they provide a easy way to "denature" drugs and make them safe to toss in the household trash. Some companies have formed partnerships with elected officials to distribute free...
Local Pharmaceutical Stewardship Laws Achieve Their Promise
When counties in California and Washington state, like Alameda and King, began enacting pharmaceutical stewardship policies several years ago it was out of the compelling need to provide more medicine take-back services to their residents. Now in 2017, the robust and...
WA Secure Drug Take-Back Act Advances Against Pharmaceutical Industry Opposition
The Washington State Legislature has an opportunity to act on a public safety bill that helps address the epidemic of opioid and medicine abuse and overdose deaths in our communities by creating a statewide medicine take-back program. The WA Secure Drug Take-back...
Kitsap and Pierce Counties Take Action on Secure Medicine Return
Two more local Boards of Health in WA state took action last week to provide their communities with more secure and environmentally sound disposal systems for leftover prescription and over-the-counter medications. On Dec. 6th, the Kitsap County Public Health board...